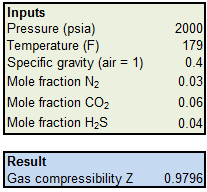
SOLVED: Question: Using the equation for the compressibility factor, Z, calculate the value of Z for water at p = 35 MPa and 500°C (where the constant for steam is R =


Physical Chemistry The Compression Factor (Z) [w/1 example

COMPRESSIBILITY factor Z, Using P and v in 3 Minutes!

EGR 334 Thermodynamics Chapter 3: Section ppt video online download

Estimate the pressure of water vapor at a temperature of $50

COMPRESSIBILITY factor Z, Using P and v in 3 Minutes!
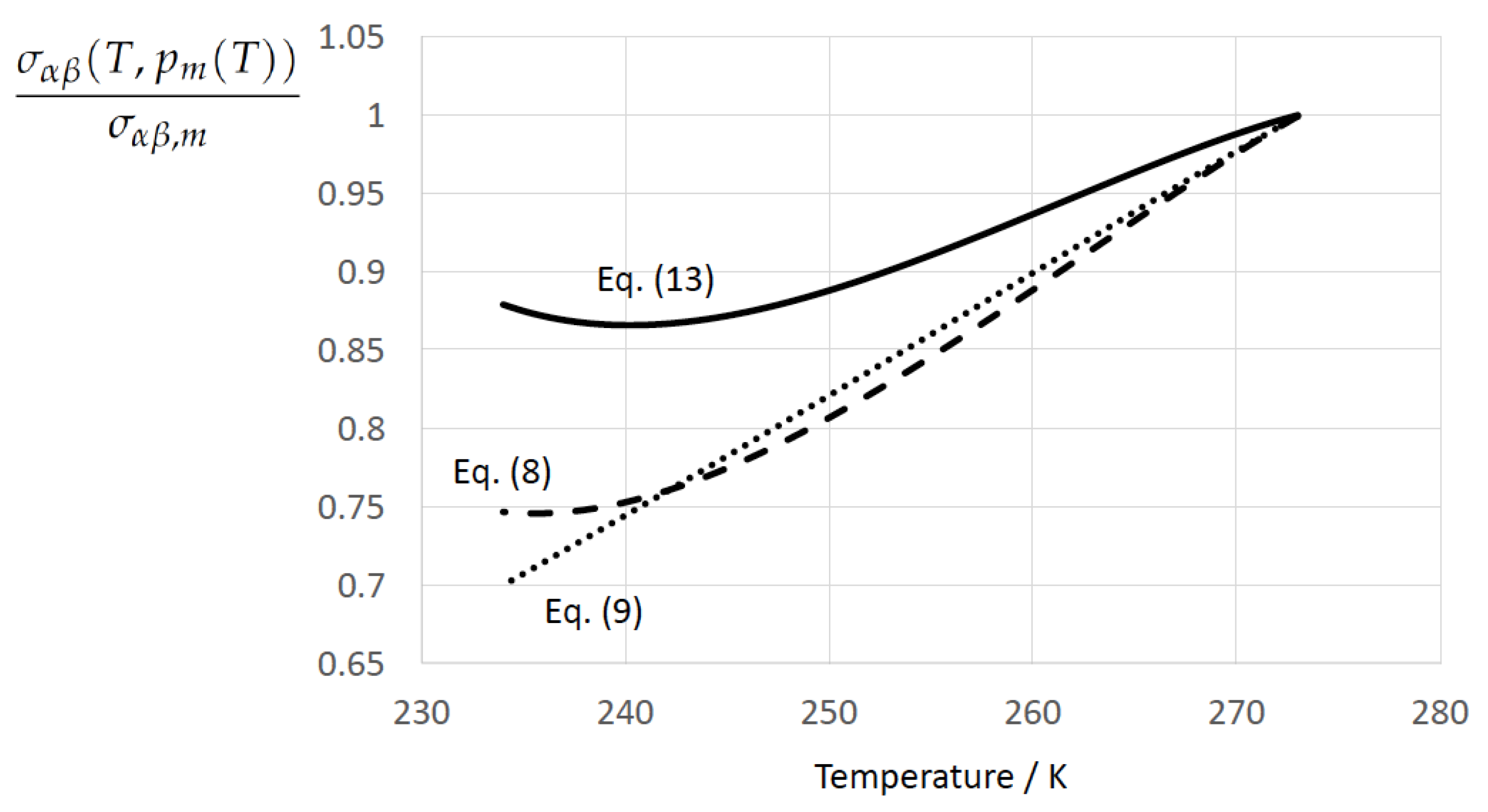
Entropy, Free Full-Text
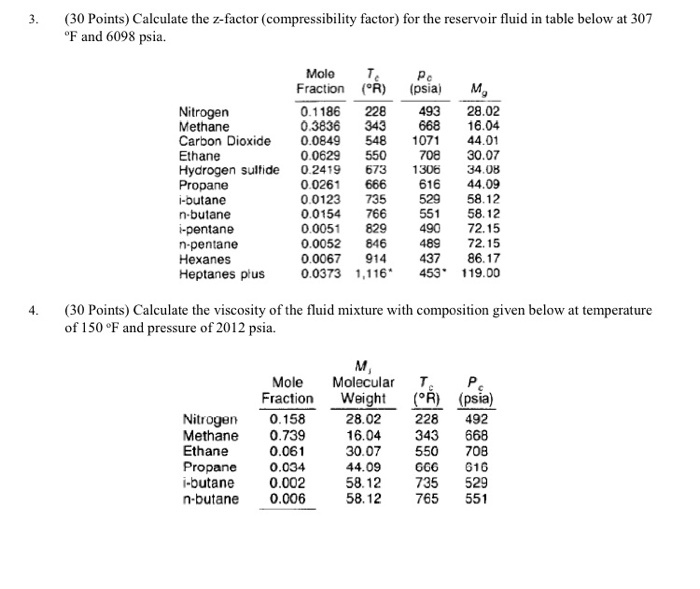
Solved 3. 30 Points) Calculate the z-factor (compressibility

3.3: Real gas and compressibility factor - Engineering LibreTexts

thermo_5th_chap03p061.pdf

If Z is a compressibility factor, van der Waals equation at low

The value of compressibility factor at the critical state the gas

3.2 Real gas and compressibility factor – Introduction to

The compressibility factor for a real gas is expressed by, z =1+ BP / RT. The value of B at 500 K and 600 bar is 0.0169 L / mol. Find the

Solved 2. (20 points) At low pressures, the compressibility